Aerobic Cellular Respiration

Both Aerobic Cellular Respiration and Anaerobic Cellular Respiration are profound topics requiring some understanding in biological chemistry.
As a part of metabolism all living things undergo Cellular Respiration. During this process, food molecules are broken down to produce energy.
Aerobic cellular respiration is often thought of as a kind of breathing, but this is not correct.
Aerobic Cellular Respiration is the process by which energy is made in all cells. As part of this process animals and humans take in oxygen (O2) and release carbon dioxide (CO2) by breathing.
Plants do not breathe as animals do and cellular respiration in plants has different pathways.
The aerobic cellular respiration equation...
C6H12O6 + 6O2 --> 6H2O + 6CO2 + Energy
meaning...
Glucose + Oxygen --> Water + Carbon Dioxide + Energy
Much more basic version of the aerobic cellular respiration equation is:
C6H12O6 + O2 --> H2O + CO2
Respiration is the process of extracting stored energy from glucose and storing it in the high energy bonds of ATP molecule. ATP (Adenosine Triphosphate) is also known as the energy currency of life, which can store and transport the energy required by all metabolic functions of living cells. As a result of aerobic cellular respiration, energy is released from the chemical bonds and used for phosphorylation of ATP. Phosphorylation is the process of adding a phosphate group to a molecule. By adding a phosphate ADP it becomes ATP. The respiration reactions are controlled by ENZYMES.
There are two types of Cellular Respiration: Anaerobic Cellular Respiration and Aerobic Cellular Respiration. Some organisms use the Anaerobic Cellular Respiration pathway, and some organisms use the Aerobic Cellular Respiration pathway.
Glossary...
Anaerobes are organisms that use the Anaerobic Cellular Respiration pathway. Most (not all) anaerobes are bacteria. Anaerobes do NOT require oxygen.
Aerobes are organisms that use the Aerobic Cellular Respiration pathway. Aerobes require oxygen.
Obligate Anaerobes oxygen is LETHAL to these organisms
Anaerobic Cellular Respiration does not require oxygen. The 2 most common forms of Anaerobic Cellular Respiration are: Alcoholic Fermentation, and Lactic Acid Fermentation.
Strict Respiration - can only carry out Respiration only one way aerobic or anaerobic.
Facultative Respiration - can switch respiration types depending on O2 availability. Example yeast.
Cells normally contain small amounts of ATP produced during the glycolysis reaction of cellular respiration. Whenever the body needs lots of energy in a hurry, its cells quickly burn this stored ATP. At this point, the cells of the bodys muscles are producing most of their ATP by lactic acid fermentation, the slow process by which glucose is converted to lactic acid. This process supplies around 2 ATP energy molecules for approximately 90 more seconds of activity. For exercise longer than 90 seconds, the cell has to complete two more reactions for energy.
These two reactions are called the Krebs Cycle and the Electron Transport Chain. These two reactions are the only way to generate a continuing supply of ATP. This is because the two reactions release energy much faster than Glycolysis and/or Lactic Acid Fermentation. For the first 15 minutes of prolonged activity, your body uses energy stored in glycogen (this is a stored form of glucose). After using the available glycogen, the body begins to burn energy stored as fat. This is one reason why aerobic exercise is so beneficial it helps you burn off that extra love.
Three Phases Of Aerobic Cellular Respiration
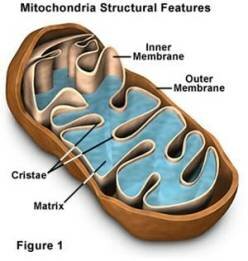
Aerobic Cellular Respiration happens in Mitochondria. Three main reactions are involved:
- Glycolosis occurs in cytoplasm of mitochondria (requires 2 ATP to start/ makes 2 ATP)
- Krebs Cycle occurs in matrix of mitochondria (makes 2 ATP)
- Electron Transport Chain occurs in mitochondria; makes majority of ATP (32 ATP)
Out of 38 ATP Produced - energy of 2 ATP required to start the process.
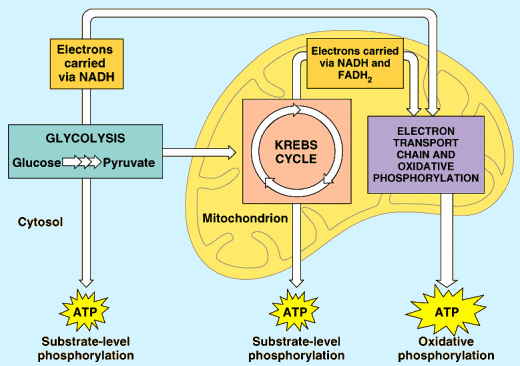
Phase 1: Glycolysis
Glycolysis takes place in the cytoplasm. In order for Aerobic Cellular Respiration to continue the Pyruvic Acid is first converted to Acetic Acid by losing a carbon atom and 2 oxygens as CO2. The Acetic acid then must enter the matrix region of the mitochondria. The CO2 produced is the CO2 animals exhale when they breathe.
So the First Stage of Respiration for ALL living organisms, anaerobes or aerobes, is called Glycolysis and takes place in the Cytosol. Glyco means glucose/sugar, and lysis means to split. Therefore means to split. Therefore, glycolysis means to split glucose This process was likely used to supply energy for the ancient forms of bacteria. Glycolysis function is to split glucose and produce NADH, ATP and Pyruvate (pyruvic acid). The process occurs in 9 steps- 6 of the steps use magnesium (Mg) as a cofactor.
For many organisms, however, glycolysis is only the beginning of the story in terms of energy production. Organisms that are able to perform aerobic cellular respiration can fully oxidize glucose and utilize more of its energy through glycolysis and three more pathways that occur after glycolysis. These three pathways, in the order that they occur, are pyruvate oxidation, the citric acid cycle, and oxidative phosphorylation.
The complete oxidation of glucose through these pathways, releases more energy than glycolysis alone. This energy, in turn, is ultimately captured in the bonds of ATP. In fact, the pathways of aerobic cellular respiration release around 34 more molecules of ATP per molecule of glucose, resulting in a total of 36 molecules of ATP per molecule of glucose 18 times the amount of energy produced by glycolysis alone!
Reactants for Glycolysis
- Glucoseas activation energy
- 4 ADP and 4P
- Enzymes
- 2 NAD+ (Nicotinamide Adenine Dinucleotide, an energy carrier)
Products of Glycolysis
- 2 Pyruvic Acids (a 3C acid)
- 4 ATP
- 2 NADH
Glycolysis - Net Result
- 2 Pyruvic Acid
- 2 ATP per glucose (4 2 = 2)
- 2 NADH
In summary, glycolysis takes one glucose and turns it into 2 pyruvates (molecules of pyruvic acid), 2 NADH and a net of 2 ATP.
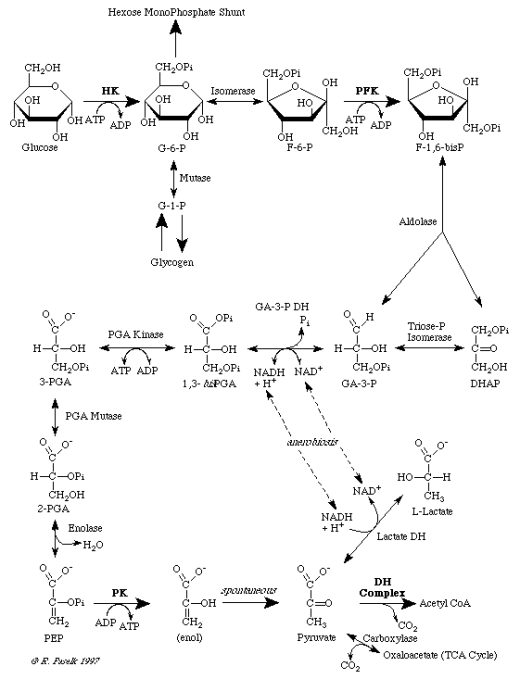
Phase 2: Steps Of The Krebs Cycle (The Citric Acid Cycle)
The pathways of aerobic cellular respiration begin where glycolysis ended with pyruvate. Pyruvate oxidation occurs at the inner mitochondrial membrane of eukaryotic cells, and at the cell membrane of prokaryotic cells. During this process, 3-carbon pyruvate is oxidized to 2-carbon acetate, which is then bound to a second molecule, called Coenzyme A. The energy-releasing oxidation reaction is coupled to the energyrequiring reduction of NAD+ to NADH + H+. The energy and electrons thus captured in NADH + H+ will play an important role later in the process...
In addition, note that one carbon molecule from pyruvate is completely oxidized and released as carbon dioxide. What does this mean in terms of our original 6-carbon glucose molecule? Each glucose molecule produces two 3-carbon pyruvate molecules. When each of these molecules goes through pyruvate oxidation, it loses one fully oxidized carbon. This means that a total of two carbons of our original glucose molecule are now fully oxidized. Two down, four to go! The product of pyruvate oxidation, called acetyl-CoA, may now enter the next pathway of aerobic cellular respiration, the citric acid cycle.
Three main steps of the krebs cycle (or the citric acid cycle) are accomplished...
- Acetyl-CoA is oxidized to produce two molecules of carbon dioxide.
- The energy released from these oxidation reactions is used to reduce the electron carriers NAD+ and FAD+ to produce NADH + H+ and FADH2, and to produce ATP by substrate-level phosphorylation.
- Citric acid cycle intermediates are rearranged to regenerate oxaloacetate, the same molecule that initiates the cycle by condensing with acetyl-CoA.
So let's take a closer look at the steps involved in this process. When acetyl-CoA enters the citric acid cycle - which is also called the Krebs cycle or the tricarboxylic acid cycle - it reacts with a 4 carbon molecule, oxaloacetate, to form a 6 carbon compound, citrate.
A series of reactions follows where citrate is rearranged. This is accompanied by the loss of a carbon atom in the form of carbon dioxide, and the formation of alfa-ketoglutarate. Oxidation of alfa-ketoglutarate results in reduction of NADH + H+, the formation of another molecule of carbon dioxide, and the production of succinyl-CoA, a 4 carbon molecule.
By this point in the cycle two molecules of reduced NADH + H+ have been formed and two molecules of carbon dioxide have been released. Succinyl-CoA enters a sequence of reactions that results in the reformation of oxaloacetate. During this part of the cycle two more molecules of NADH + H+ and FADH2 are produced, and ATP is formed. This completes the cycle.
Now, let's step back and try to see the big picture. First, a four carbon molecule and a two carbon molecule are combined to form a six carbon molecule. Second, two carbon atoms are completely oxidized, producing 2 molecules of carbon dioxide. The energy produced from the oxidation reactions of this cycle is captured in the bonds of ATP, NADH + H+ and FADH2. Third, there is a series of reactions that regenerates the original four carbon molecule that started the cycle.
Once the Acetic Acid enters the Matrix it combines with Coenzyme A to form a new molecule called Acetyl-CoA. The Acetyl-CoA then enters the Krebs Cycle. This phase produces most of the cell's energy in the form of NADH and FADH2, not ATP and does NOT require O2. CoA breaks off to gather more acetic acid. The Acetic acid is broken down. Here is the Krebs Cycle diagram...
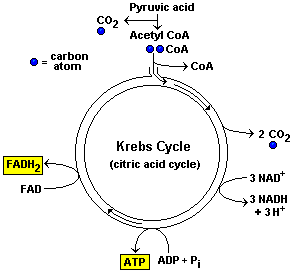
Result of one turn of the Krebs cycle...
As a result of one turn of the Krebs cycle the cell makes:
- 1 FADH2
- 3 NADH
- 1 ATP
However, each glucose produces two pyruvic acid molecules . So the total outcome is:
- 2 FADH2
- 6 NADH
- 2 ATP
Phase 3: Oxidative Phosphorylation and The Electron Transport Chain
The process extracts energy from NADH and FADH2 in order to add a phosphate group to ADP to make ATP. It occurs in Mitochondria cristae. Oxidative Phosphorylation Requires NADH or FADH2 ADP and PO2. Oxidative phosphorylation Requires the Electron Transport Chain. The Electron Transport Chain is a collection of proteins, embedded in the inner membrane. It is used to transport the electrons from NADH and FADH2.
Glossary...
Cytochrome C is one of the proteins of the electron transport chain which exists in all living organisms and often is used by geneticists to determine relatedness.
Chemiosmotic Hypothesis. Biologists still dont know exactly how ATP is made. The best theory we have is called the Chemiosmotic Hypothesis. proposes that the Electron Transport Chain energy is used to move H+ (protons) across the cristae membrane, and that ATP is generated as the H+ diffuse back into the matrix through ATP Synthase.
ATP Synthase uses the flow of H+ to make ATP. It works like an ion pump in reverse, or like a waterwheel under the flow of H+ water.
So the final stage of aerobic cellular respiration is oxidative phosphorylation. The key event of this stage is the formation of a proton gradient that drives the synthesis of ATP. Heres how it happens. In the first step, an electron carrier (such as NADH + H+) donates electrons to the first protein in the chain, resulting in the protein becoming reduced.
This is followed by the sequential oxidation and reduction of a chain of proteins and other molecules that comprise the electron transport chain. These events are accompanied by the transfer of protons across the membrane. The energy needed to pump these protons across the membrane is derived from the redox reactions that occur.
The electron transport chain consists of several proteins and other compounds that are associated with the inner membrane of mitochondria, or the cell membrane of prokaryotes. In the first step, the electron carrier NADH donates electrons to the first protein in the chain the NADH-Q reductase complex. Once NADH-Q reductase is reduced, it can in turn become oxidized by reducing the next component of the chain, ubiquinone.
Ubiquinone then may become oxidized by reducing the next component of the chain, and so on. Note that each component of the chain undergoes a cycle of being reduced and then oxidized as it gains and then donates electrons. At the end of the electron transport chain, the electrons are finally used to reduce the terminal electron acceptor, which in aerobic metabolism is oxygen. The reduction of oxygen yields water - the final product of this process.
At this point we have finally found where oxygen takes part in aerobic cellular respiration, or, in other words, why aerobic cellular respiration is aerobic. In fact, this is the only step in aerobic cellular respiration that directly requires molecular oxygen. If oxygen is not present, however, oxidative phosphorylation, the citric acid cycle, and pyruvate oxidation are not able to occur. We will discuss the reason for this in the lesson on fermentation.
The sequence of reduction and oxidation reactions allows energy to be gradually released, and it is productively used to transport protons (H+) across the membrane. The transport of protons requires energy because it is accompanied by the formation of a chemical gradient. As weve seen before, a chemical gradient is a form of potential energy remember this energy, well see how it is used to produce ATP in just a moment.
Electrons can actually enter the electron transport chain at several stages in the process. Precisely where they enter depends on the redox potential of the electron donor and the redox potential of the electron acceptor (if this doesnt make sense to you, then it might be useful to review basic redox chemistry in a chemistry textbook).
As you recall, in the citric acid cycle FAD+ is reduced to form FADH2. The electrons carried by FADH2 enter the electron transport chain at ubiquinone. From this point on, the electrons follow the same path as with NADH + H+ and are ultimately used to reduce oxygen. Two other important features of electron transport are notable.
First, notice that each time a redox reaction occurs, the amount of free energy present decreases. In other words, each time an electron is transferred between carriers, a small amount of energy is released. This, again, is the energy that is used to build a proton gradient across the membrane. Second, it is important to notice that all components of electron transport chains share a common feature - they are all capable of undergoing oxidation and reduction.
As a result, they cycle from being oxidized, to being reduced, to being oxidized, and so on. This has several important implications, one of which is that the NADH + H+ produced during metabolism of glucose is continually being re-oxidized to form NAD+ that can cycle back to participate in glycolysis, pyruvate oxidation, and the citric acid cycle.
Like many other membranes in cells, the inner mitochondrial membrane is highly selective and H+ cannot readily cross it. This is a very important feature since it provides a way to form a H+ gradient. During electron transport, protons are pumped across the membrane via the electron transport chain, forming just such a gradient.
Given a chance, protons will diffuse back across the membrane toward equilibrium. In order for H+ to move back across the membrane, however, they must pass through special channels within the ATP synthase complex. The passage of H+ through the channels is driven by the electrochemical gradient that exists due to the difference in the H+ concentrations on either side of the membrane.
This electrochemical gradient, which is also called the proton motive force, provides the energy needed by ATP synthase to synthesize ATP from ADP and inorganic phosphate. As protons flow down their electrochemical gradient and through ATP synthase, they cause part of the synthase complex to rotate. The energy thus provided is used to form ATP from ADP and inorganic phosphate.
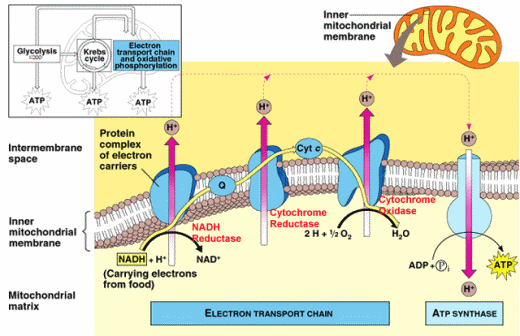
ATP Summary...
To summarize the process of aerobic cellular respiration, lets look again at how ATP is produced throughout the whole process. Note that only a small proportion of ATP is produced during glycolysis and the citric acid cycle - these processes only net 4 ATP.
The reduced electron carriers NADH + H+ and FADH2 cycle energetic electrons to the electron transport chain from glycolysis, pyruvate oxidation, and the citric acid cycle. The vast majority of the ATP is produced as a result of oxidative phosphorylation.
In fact, 32 (!) molecules of ATP are produced for each molecule of glucose that is metabolized. The yield of energy from aerobic cellular respiration is important since many cells need a lot of ATP to function and grow. Just how much ATP do cells need? Heres a staggering example: a working muscle cell may recycle ATP at a rate of 10 million molecules per second! Maybe sugar isnt so bad for you after all!
Link to this page, insert the following code into your web page/blog...